Citation: Shaked T, “Evolution of an On-Body Injector: from Drug-Specific Device to Platform Technology”. ONdrugDelivery Magazine, Issue 100 (Sep 2019), pp 60-63.
Tsachi Shaked introduces E3D’s On-body Bolus Injector, OBI, a versatile wearable injector platform that uses gas generating microcells as its engine, has no electromechanical components, only one moving part, and can be supplied to the patient prefilled and preloaded or filled and loaded at the time of application.
“The first version of the OBI, the OBI-1 provides subcutaneous delivery of a 0.6 mL dose of pegfilgrastim in the familiarity, comfort and convenience of the patient’s home, saving them the inconvenience and discomfort of yet another hospital visit.”
Some of medicine’s greatest innovations started with a simple question, “What if?” What if tiring hospital visits could be completely eliminated by creating an on-body injector which could allow patients to receive vital drug dosages at home?
As the trend to transition patient care from the clinic to the home continues, and as a growing number of parenteral biologic products are developed for a wide range of medical conditions, there is an ever greater need for devices which enable patients or other non-clinicians to administer injectable medications themselves, safely and effectively.
MOVING CARE FROM THE PATIENT CHAIR TO THE SOFA
Elcam Drug Delivery Devices (E3D), a division of the Elcam Medical Group, is at the forefront of moving elements of patient care out of the clinic and into the home, where it is appropriate and safe to do so. The move into on-body bolus injectors was a natural progression for E3D, an established player in the injectable medications market (see Figure 1) and best known for its Flexi-Q PFS disposable mechanical autoinjector which is partnered with a big pharma “blockbuster” drug.
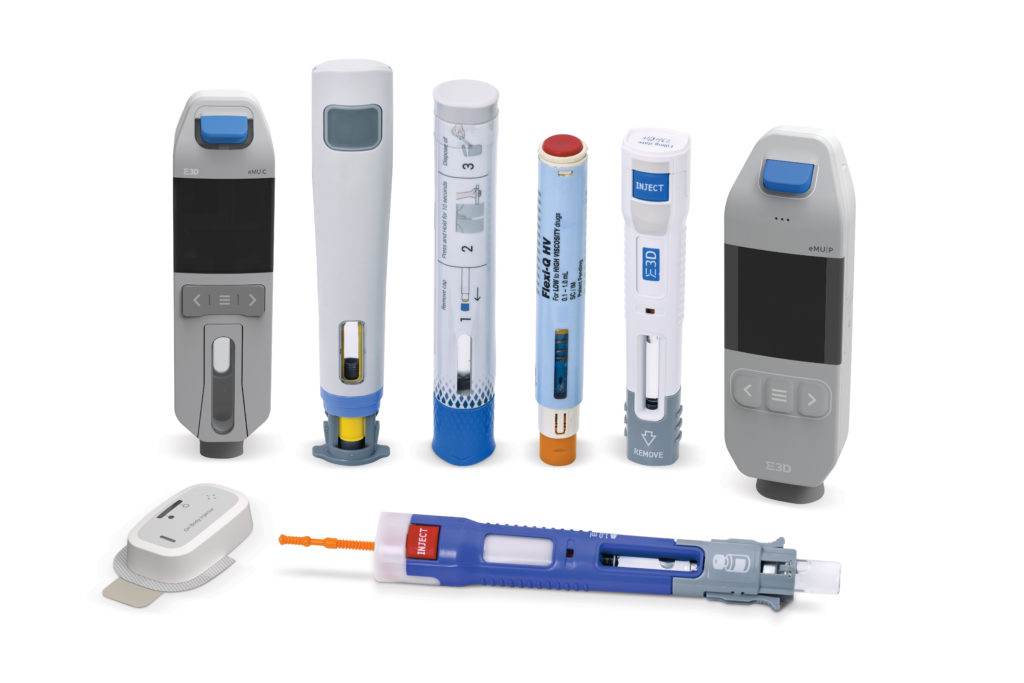
Figure 1: E3D’s family of products.
MARKET DRIVERS
In recent years there has been considerable interest in on-body injectors from pharma companies developing biosimilars of Amgen’s Neulasta (pegfilgrastim). This drug is delivered as a single 0.6 mL subcutaneous injection one day after a course of chemotherapy, in order to prevent neutropenia and therefore reduce risk of infection. Amgen’s Onpro on-body injector device was adapted from an insulin pump technology. If an insulin pump delivers too much insulin too quickly, it can result in the user entering into a hypoglycaemic condition – which could lead to serious injury or death. Consequently, an insulin pump must have the appropriate means to deliver very small (but variable) amounts of insulin, very accurately, over extended periods – with associated sophisticated monitoring and control systems to ensure clinical efficacy and user safety.
“In addition to avoiding the need for complex and expensive motors, gears and their associated power sources and controls, the use of such microcells in this way results in a device technology which is small, light, inherently robust, with only one moving part.”
By contrast, one-time delivery of a 0.6 mL bolus of pegfilgrastim takes place over a period of minutes. While the drug must undoubtedly be delivered accurately and safely, this does not present the same level of challenge as delivery of insulin – and can therefore be achieved with a device that is smaller, less sophisticated and less costly than a modified insulin pump or other relatively complex electromechanical technology.
I was intrigued by this possibility, as was our development team. Creating on-body delivery devices represented the next step in E3D’s drive to solve injection-related challenges by delivering suitable device platforms for subcutaneous or intramuscular delivery of medications outside of a clinical environment. Beyond the strong business case, I had a personal motivation. I was driven by the words of a close friend suffering from skin cancer who said, “Wouldn’t it be nice if I could do that simple shot in my own home?”
Patients taking pegfilgrastim are already weakened from chemotherapy. Creating a convenient injector for home use spares them the tiring trip to the hospital, finding a parking space, and waiting in line. Instead, they can rest comfortably at home while the on-body injector does its job. It also spares valuable hospital resources, providing a cost-effective alternative to outpatient treatment.
TOWARDS AN ON-BODY PLATFORM TECHNOLOGY
While working on the new on-body device, the E3D team looked at several key issues including the device’s size, relative complexity and estimated manufacturing costs. Our research determined that reliable and robust on-body devices for drug delivery should ideally meet the following criteria:
- Usability – replicate the user experience already introduced to market, ensuring it is both easy to use and user-friendly
- Pre-loading – the device can be loaded with the drug by hand from a PFS and applied to the patient’s skin by a healthcare professional on the day of the patient’s last clinic visit for chemotherapy
- Delivery – the bolus injection will be administered within a specified time, automatically starting 27 hours after being applied to the patient’s skin
- Stable – doesn’t protrude too much from the patient’s skin and cannot be easily dislodged from a patient’s body in the 27 hours between device placement and injection
- Reduces risk – if the device is not properly attached to the patient or the intended site of injection has to be changed because a nerve end or blood vessel had been hit by the injection cannula, it will not result in losing the drug dosage
- Simplified design – reduce the size and complexity of the device to enable cost-effective manufacture.
BOLUS SC DELIVERY WITH ON-BODY INJECTORS

Figure 2: OBI on-body injector.
Following extensive R&D and usability testing, E3D recently introduced its On-body Bolus Injector (OBI), which met all the defined criteria. The OBI platform (Figure 2) has been specifically developed to deliver bolus subcutaneous injections at the desired injection rates — as cost-effectively as possible and with minimal need for patient intervention. Drawing on E3D’s extensive experience in the design of wearable, smart and mechanical injectors, the first version of the OBI, the OBI-1 provides subcutaneous delivery of a 0.6 mL dose of pegfilgrastim in the familiarity, comfort and convenience of the patient’s home, saving them the inconvenience and discomfort of yet another hospital visit.
OBI-1 is configured to be used in a similar manner to the Onpro device which is already approved and used with the originator’s Neulasta product. OBI-1 for pegfilgrastim is manually filled from a prefilled syringe by a healthcare professional in the clinic and is then attached to the patient by an integrated adhesive patch. As with the Neulasta Onproinjector device, OBI-1 incorporates a timer which results in the patient automatically receiving their bolus injection over approximately one hour – 27 hours following attachment of the device and after the patient has returned home from the clinic.
COMPACT, ECONOMICAL AND EFFICIENT TECHNOLOGY PLATFORM
“Because OBI has no
electromechanical
components, and has
only one moving part,
it represents a device
platform which is
compact, inherently
robust, economical, readily
scalable for any dose
volume, and suitable for
either manual filling of
drug or preloaded with
the relevant drug.”
The key to the OBI technology platform is its use of gas generation by microcells as the basis for its “engine” – a system which is well-established to move liquids and deliver fluids in numerous medical and consumer products. Once an OBI device has been activated, gas is automatically generated at a controllable rate and its pressure is utilised to move a plunger and deliver the bolus of drug. In addition to avoiding the need for complex and expensive motors, gears and their associated power sources and controls, the use of such microcells in this way results in a device technology which is small, light, inherently robust, with one moving part (the elastomeric plunger which delivers the drug) and very cost-effective.
The OBI device technology platform offers significant advantages in the delivery of other injectables. Because OBI has no electromechanical components, and has only one moving part, it represents a device platform which is compact, inherently robust, economical, readily scalable for any dose volume, and suitable for either manual filling of drug or preloaded with the relevant drug.
Whilst the OBI-1 is designed specifically for the delivery of a 0.6 mL dose of pegfilgrastim, and for manual filling of its reservoir by a healthcare professional, E3D recognises that application of the OBI platform (Figure 3) for other drugs will typically require injection of higher dose volumes. An OBI device with a single microcell is capable of delivering a liquid bolus of up to 20 mL, allowing OBI devices to be readily scaled for such dose volumes. Should it be necessary to administer even higher dose volumes, additional microcells can be easily incorporated as necessary. The microcell “engine” is also well suited to delivering viscous liquids, addressing an increasingly prevalent requirement for emerging drug products.

Figure 3: OBI platform technology.
PRELOADED DEVICES WILL OFFER SIGNIFICANT PATIENT ADVANTAGES
Additionally, and in common with other injector devices, there are clear advantages in having an injector which is intended for use by patients to be supplied with the drug product already preloaded within the device – in order to make its use as simple and intuitive as possible. Whilst OBI-1 for pegfilgrastim is designed for manual filling, the simplicity of the OBI platform allows E3D to offer OBI devices in a configuration that incorporates prefilled primary containers (in glass or other materials), which are suited to established drug filling, handling, sterilisation and assembly processes.
Figure 4: Wireless connectivity.
While OBI-1 for pegfilgrastim incorporates a timer to enable automatic activation and initiate an injection 27 hours post-attachment of the device to the patient, it is not anticipated that this feature will be widely required for other drugs and indications, with OBI devices typically therefore being activated by the user pressing a button following placement of the device on the injection site tissue. Once activated, the OBI device communicates with the patient by means of audible and visual indicators, making it clear when an injection has been completed and when the used device can be peeled from the skin for disposal. Since it generates no electromechanical waste, OBI can be disposed of simply and without the need for complex and expensive “back to base” logistical operations.E3D’s technology portfolio includes “smart” injector devices which are configured for generation, storage and wireless communication (e.g. to central data storage and/or mobile phones) of comprehensive data regarding each injection (Figure 4).
The OBI platform can provide the required capabilities to meet the needs of specific drugs, therapies and patient populations. Its versatility is set to provide significant added value to pharma companies seeking cost-effective on-body alternatives.
EXPANDING THE BOUNDARIES OF ON-BODY TECHNOLOGY
With more injectables requiring high dose volumes and with more of these drug products having elevated viscosities, all of the signals from the marketplace suggest that on-body injector devices will have an increasingly important role to play in providing, effective, safe and convenient patient care.
While OBI-1 represents a compact, robust, flexible and cost-effective on-body injector for delivery of pegfilgrastim, devices based on E3D’s OBI technology platform create significant advantages for a broad range of drugs for a wide variety of indications including autoimmune conditions, diabetes, oncology, multiple sclerosis and more.
On-body injectors will represent an increasingly attractive alternative to handheld injector devices for higher dose volumes and viscous drugs. Our goal is to provide a robust, scalable and economic platform that serves many applications.



