Citation: Fan J, “I-Platform Device: The Smart Device with Aptitude”. ONdrugDelivery, Issue 117 (Mar 2021), pp 76–77.
Jimmy Fan outlines the capabilities and benefits of the I-Platform smart drug delivery device.
The dream of many pharma and biotech companies is to have a device that is capable of integrating with as many drug containers as possible – and functions as an Apple like device that is easy to use, friendly, yet powerful and economic.
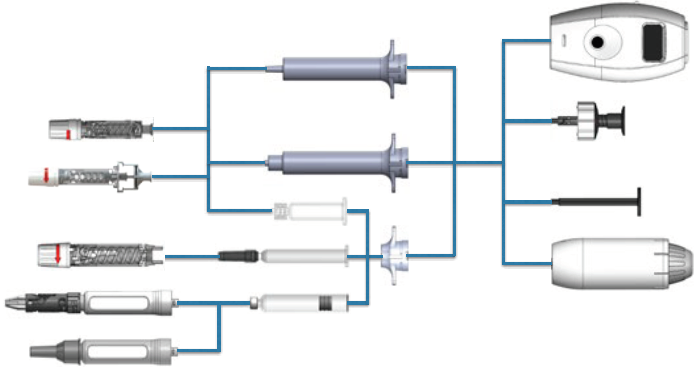
Figure 1: The I-Platform drug delivery device – showing the safety needles on the left, the containers and their adapters in the middle and the delivery units on the right.

Figure 3: The Beta safety pen needle.

Figure 2: Prefilled
syringe with Ascpo
safety needle.
It can seem like too much to ask – yet CCBio treats this unmet need as a goal. It has become part of the company’s everyday work, with the eventual aim of introducing the I-Platform device (Figure 1) to industries and users. The concept is simple – with one delivery unit, the I-Platform can adapt to cartridges or prefilled syringes and combine with either safety mechanisms or conventional needle shields. With the versatility of the Ascpo safety needle (Figure 2) and Beta safety pen needle (Figure 3) from CCBio, the I-Platform makes drug delivery safer and easier (Figure 4), whilst ensuring the necessary precision.

Figure 4: Adapter system (A) and safety needle (B).
Versatility is one key benefit of the adaptable I-Platform. From many years’ experience, CCBio understands the differences in dimensions and geometry between drug containers, depending on the manufacturers. These differences, although usually minor, can cause problems when it comes to functionality. For example, a difference of a few millimetres in the length of a cartridge may affect dose accuracy.
The I-Platform device (Figure 5) is very easy to fabricate and design changes can be implemented rapidly and economically because it is just one component and injection mould. During the preliminary study of a drug container, CCBio is able to detect and correct any dimension issues and secure the final product functionality and performance from the very early stage of a project, which helps lead to successful design validation testing and outcomes. Such an approach also saves money.

Figure 5: The I-Platform drug delivery device.
The delivery unit at the heart of the I-Platform is state-of-the-art technology, incorporating Wi-Fi, Bluetooth and a micro processing data system with a multifunctional LED display. Many other connectivity and smart capabilities can also be included and tailor-made for each specific requirement. The driving and power unit is extremely robust yet precise. And the device can handle viscosity up to 70 cP and dosage down to 0.1 mL, as well as allowing the injection speed and injection time to be programmed.
The result is that patient pain is reduced to a minimum to give a pleasant drug administration experience. Also, healthcare providers can reduce the risk of occupational injury from handling the injection of high-viscosity drugs. Making patients and healthcare providers love this device is the ultimate goal of CCBio. The company does not compromise the quality of the device – sourcing all the parts and important modules like the motor, battery, PCB and driving parts from local or Japanese suppliers. In addition to the electronic power unit station, CCBio can also offer manual and traditional spring driven power unit sets – giving customers the flexibility and diversity they need.
CCbio is a 100% Taiwanese biotech and medical device company, which is a one-stop-solution company. It aims to satisfy customers’ every need and resolve difficulties during product development, creating innovative products and devices and differentiating customers’ products in the marketplace.

