To Issue 152
Citation: Hasegawa H, Suzuki T, “New Extractables Data for OXYCAPT™ Multilayer Plastic Vial for Biologics & Gene/Cell Therapy”. ONdrugDelivery, Issue 152 (Oct 2023), pp 104–108.
Hiroki Hasegawa and Tomohiro Suzuki review the properties of OXYCAPT™, Mitsubishi Gas Chemical’s multilayer material for primary biologic packaging, and share the data from the latest study on OXYCAPT’s extractables profile.
“OXYCAPT is a multilayer plastic vial developed by MGC that offers a number of advantageous qualities as a primary drug container.”
INTRODUCTION
Mitsubishi Gas Chemical (MGC) is a leading company in the field of specialist polymers with oxygen-absorbing or barrier functions. In 2019, MGC launched OXYCAPT™ multilayer plastic vial & syringe, which offer excellent oxygen and ultraviolet barrier properties. Since then, a lot of biologics and gene/cell therapy companies have evaluated OXYCAPT for their products. Based on these customers’ requests, MGC has conducted new extractables studies, the results of which will be discussed in the latter half of this article.
In recent news regarding OXYCAPT syringe, MGC signed a letter of intent with Becton Dickinson (BD) in May 2022 and has started earnest discussions to apply its multilayer technology to next-generation prefilled syringes for biologics. Therefore, MGC has tentatively stopped introducing the current version of OXYCAPT syringe to customers. MGC believes this collaboration will be helpful for pharmaceutical companies to develop novel and sensitive future drugs safely.

Figure 1: OXYCAPT multilayer plastic vial.
OXYCAPT is a multilayer plastic vial developed by MGC that offers a number of advantageous qualities as a primary drug container (Figure 1). MGC continuously conducts tests to confirm OXYCAPT’s excellent properties, including:
- Excellent oxygen and ultraviolet (UV) light barrier
- Strong water vapour barrier
- Very low extractables
- High pH stability
- Low protein adsorption and aggregation
- High transparency
- High break resistance
- Easy disposability
- Lightweight material.
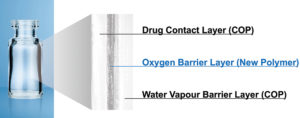
Figure 2: Multilayer structure of OXYCAPT.
OXYCAPT OVERVIEW
OXYCAPT consists of three layers – the drug contact layer and the outer layer are made of cyclic-olefin polymer (COP), and the oxygen barrier layer is made of MGC’s novel polyester (Figure 2). One variety of OXYCAPT, OXYCAPT-P, provides an excellent oxygen barrier. For example, the oxygen barrier of an OXYCAPT-P vial is about 20 times better than that of a COP monolayer vial (Figure 3).
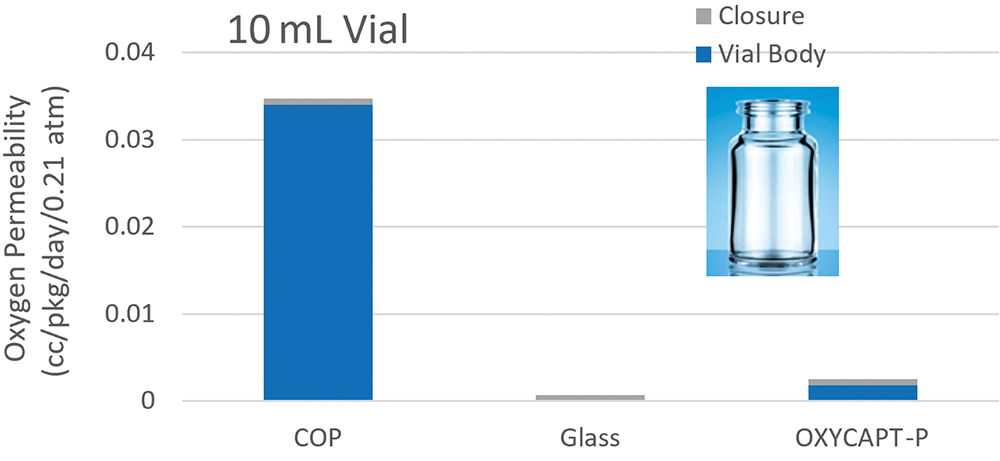
Figure 3: Oxygen permeability comparison of a typical COP, glass and OXYCAPT-P.
“While about 70% of 300 nm UV light transmits through glass and COP, only 1.7% transmits through OXYCAPT.”
MGC recently obtained a report on the environmental impact of glass and plastic containers for medical use from a Japanese research company. The report shows that plastic containers for medical use are much more environmentally friendly compared with glass containers. For example, the carbon footprint, nitrogen oxides emissions, sulfur oxides emissions and water consumption associated with plastic containers for medical use are several times smaller than those of their glass equivalents.
Furthermore, OXYCAPT provides an excellent UV barrier. While about 70% of 300 nm UV light transmits through glass and COP, only 1.7% transmits through OXYCAPT (Figure 4). MGC has confirmed that this feature contributes to the stability of biologics.
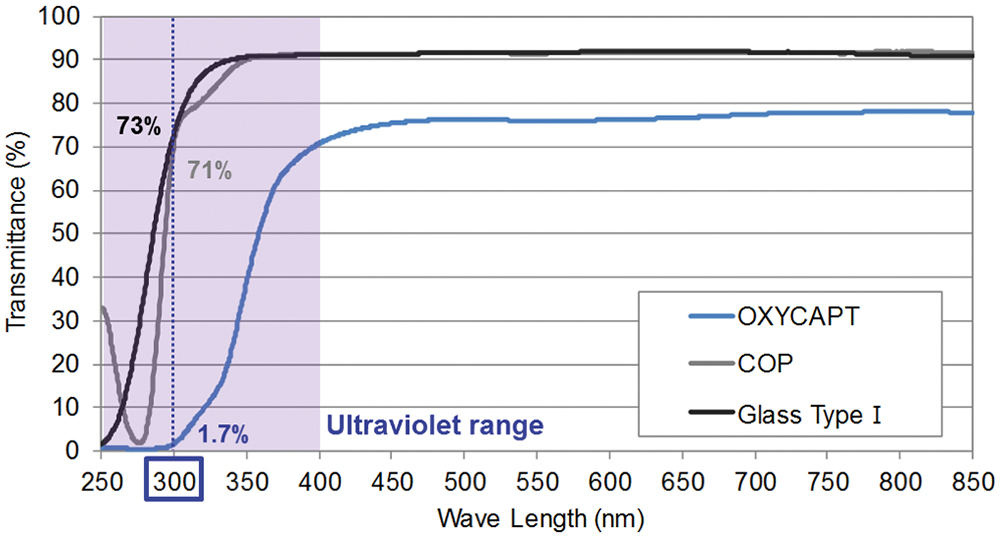
Figure 4: UV light transmittance comparison of a typical COP, Type 1 glass and OXYCAPT.
While OXYCAPT cannot reach the performance of glass with respect to acting as a water vapour barrier, its properties are similar to those of COP, which has been used for injectable drugs for a long time. This means that OXYCAPT easily meets the requirements of a water vapour barrier set out by the ICH guidelines.
“The primary target market for OXYCAPT is the therapeutic application of biologics.”
The OXYCAPT vial is produced by co-injection blow-moulding technology. MGC has also developed inspection methods for testing the oxygen barrier layer. All the containers are fully inspected by state-of-the-art inspection machinery.
Each polymer meets the requirements of US Pharmacopeia (USP) regulations USP <661>, USP <87> and USP <88>, as well as those of the European Pharmacopoeia, and has been filed in the US FDA’s drug master file (DMF). The vials are also compliant with each pharmacopoeia and have been filed in the DMF.

Figure 5: OXYCAPT plastic vial in a standard nest and tub format.

Figure 6: OXYCAPT plastic vial in a standard tray format.
MGC can offer bulk vials and ready-to-use (RTU) vials, with its RTU products provided in standard nest and tub (Figure 5) or tray (Figure 6) formats. The nest and tub are mainly sterilised using gamma rays. There are 2, 6, 10 and 20 mL variants for vials (Table 1). MGC is willing to provide samples for initial testing free of charge.
| ISO 8362-1 Vial | Length (mm) |
Width (mm) |
Outer Diameter of Flange (mm) |
Inner Diameter of Flange (mm) | Packaging Option |
| 2R (2 mL) | 35 | 16 | 13 | 7 | Nest or Tray |
| 6R (6 mL) | 40 | 22 | 20 | 12.6 | Nest or Tray |
| 10R (10 mL) | 45 | 24 | 20 | 12.6 | Nest or Tray |
| 20R (20 mL) | 55 | 30 | 20 | 12.6 | Nest or Tray |
Table 1: MGC’s OXYCAPT product portfolio.
“The results of the analyses with HS-GC-MS, GC-MS and LC-UV-MS all showed no organic compound exceeded the AET with the OXYCAPT CCS.”
The primary target market for OXYCAPT is the therapeutic application of biologics. As mentioned in ICH Q5C (Stability of Biotechnological/Biological Products), oxidation is one of the causes of protein instability. As such, the oxygen and UV barrier properties of OXYCAPT will definitely contribute to the stability of biologics stored within. Furthermore, some drug developers have recently started evaluating the OXYCAPT vials for their gene and cell therapies; the RTU vial is sterilised by gamma radiation, making it ideal for protein-based drugs.
NEW EXTRACTABLES DATA
The latest studies conducted by MGC have shown an outstanding characteristic of OXYCAPT – extremely low levels of extractables. An organic and elemental extractables study, performed in collaboration with an outsourcing pharmaceutical company, was conducted using MGC’s container closure system (CCS) comprised of a 20 mL OXYCAPT vial sterilised with 40 kGy gamma rays and a normal butyl rubber stopper.
In order to detect and quantify organic analytes, volatile compounds were measured by HS-GC-MS (headspace gas chromatography-mass spectrometry). Moreover, non-polar, volatile and semi-volatile compounds were measured by GC-MS, whereas LC-UV-MS (liquid chromatography-ultraviolet spectroscopy/mass spectrometry) was used to detect compounds with varying polarity and volatility. Furthermore, levels of the elemental impurities specified in ICH Q3D plus aluminium and tungsten were detected and quantified by inductively coupled plasma mass spectrometry (ICP-MS).
These studies were conducted by a 48-hour incubation at 50°C of the OXYCAPT CCS with four different formulations:
- 0.1% aqueous polysorbate 20 (PS-20)
- 0.1 M phosphate buffer at pH 3
- 0.1 M phosphate buffer at pH 10
- 50% ethanol solution.
These extraction solutions cover those outlined in the latest draft of USP <665>. After incubation, a sample work-up for each solvent and analysis method was performed. It was then confirmed whether or not there were any organic extractables that exceeded the analytical evaluation threshold (AET). Each AET of HS-GC-MS, GC-MS and LC-UV-MS was determined by calculation with respect to the safety concern threshold (1.5 μg), filling volume of the vial, maximum number of vials administered per day and the concentration factor due to sample work-up. The results of the analyses with HS-GC-MS, GC-MS and LC-UV-MS all showed no organic compound exceeded the AET with the OXYCAPT CCS (Table 2).
| Analytical method |
Solution | Sample work-up |
AET (ppm) |
Compounds found above AET |
| HS-GC-MS | 0.1 % PS-20 | Direct | 0.015 | No peaks were detected at levels above the AET |
| pH 3 buffer | Direct | |||
| pH 10 buffer | Direct | |||
| GC-MS | 0.1 % PS-20 | Extraction | 0.3 | No peaks were detected at levels above the AET |
| 50% EtOH | Extraction | |||
| pH 3 buffer | Extraction | |||
| pH 10 buffer | Extraction | |||
| 50% EtOH | Evaporation | |||
| LC-UV-MS | 0.1 % PS-20 | Extraction | 0.3 | No peaks were detected at levels above the AET |
| 50% EtOH | Extraction | |||
| pH 3 buffer | Extraction | |||
| pH 10 buffer | Extraction | |||
| 50% EtOH | Evaporation |
Table 2: Organic extractables profile of OXYCAPT.
“These latest results have contributed to the ongoing studies verifying OXYCAPT’s superior properties for biologics and gene/cell therapies.”
In the study with ICP-MS, no elemental impurities exceeded their permitted daily exposure (PDE) as outlined in ICH Q3. Aluminium and tungsten, which are not listed in ICH Q3D, were also not present at a significant level (Table 3). This result indicates that OXYCAPT can contribute to a safety assessment for drugs and the pH stability of drug solution.
| Element | 0.1% PS-20 pH 10 buffer |
pH 3 buffer | PDE (μg) |
| Amount administered with 5 vials (μg) | |||
| Ag | < 1.50 | < 1.50 | 15 |
| As | < 1.50 | < 1.50 | 15 |
| Au | < 30 | < 30 | 300 |
| Ba | < 70 | < 70 | 700 |
| Cd | < 0.2 | < 0.2 | 2 |
| Co | < 0.5 | < 0.5 | 5 |
| Cr | < 110 | < 110 | 1100 |
| Cu | < 30 | < 30 | 300 |
| Hg | <0.3 | <0.3 | 3 |
| Ir | <1 | <1 | 10 |
| Li | <25 | <25 | 250 |
| Mo | <150 | <150 | 1500 |
| Ni | 2 | <2 | 20 |
| Os | <1.00 | <1.00 | 10 |
| Pb | <0.50 | <0.50 | 5 |
| Pd | < 1.00 | < 1.00 | 10 |
| Pt | < 1.00 | < 1.00 | 10 |
| Rh | < 1.00 | < 1.00 | 10 |
| Ru | < 1.00 | < 1.00 | 10 |
| Sb | <9 | <9 | 90 |
| Se | <8 | <8 | 80 |
| Sn | <60 | <60 | 600 |
| Ti | <0.8 | <0.8 | 8 |
| V | <1 | <1 | 10 |
| Al | <6 | <20 | n.a |
| W | < 20 | < 20 | n.a |
Table 3: Elemental extractables profile of OXYCAPT.
CONCLUSION
In conclusion, these latest results have contributed to the ongoing studies verifying OXYCAPT’s superior properties for biologics and gene/cell therapies. In addition to the advantages of COP, such as a strong water vapour barrier, high break resistance, very low extractables and low protein adsorption, OXYCAPT also provides a strong oxygen and UV light barrier. MGC believes that OXYCAPT offers a multitude of benefits to the rapidly growing field of biologics and gene and cell therapies.
Learn more about Mitsubishi Gas Chemical’s OXYCAPT™ at: www.mgc.co.jp/eng/products/abd/oxycapt.


