Citation: “Injecting High Volume, High Viscosity Drugs”. ONdrugDelivery Magazine, Issue 83 (Feb 2018), pp 6-8.
SHL has extensive experience in the parenteral arena. In this article, SHL details its latest offerings in the world of biologics, as well as its partnership with QuiO, a speciality digital healthcare start-up, to develop injectables in the connected healthcare space.
The rising trend towards biologics in the pharma industry has opened the gates for the development of very specific molecules targeting the diseases right at their core. Due to their effectiveness, the market for biologics and biosimilars is expected to grow at an annual rate of over 8%.1 We now see several disease areas, historically dominated by a few blockbuster products, splitting up into a number of different sub-diseases, each with their own tailored treatments.
Upon first inspection, this appears to be a huge win-win scenario, but it comes with a number of challenges. As diseases are split up into sub-categories, the patient groups naturally become smaller. Therefore, to maintain returns on investment, higher prices and/or decreased R&D costs must be implemented. At the same time, there is a focus on patient convenience and self-treatment has become the gold standard in subcutaneous injections. Also worth noting is a strong trend towards less frequent administration and, as a result, increasingly large dosing.
“As diseases are split up into sub-categories, the patient groups naturally become smaller. Therefore, to maintain returns on investment, higher prices and/or decreased R&D costs must be implemented…”
With autoinjectors delivering higher doses of proteins subcutaneously, there is the major consideration of injection duration. A duration of 10-15 seconds has historically been regarded as the maximum acceptable threshold for autoinjection – patients typically prefer to not hold a device in place for any longer.
The complex molecular structure of biologics also poses a technical challenge. Biologics have a propensity towards destabilising or aggregating in a prefilled syringe or other primary package over the course of their shelf life. They often need to be delivered in higher concentrations to have an effect, and with increasing concentration the viscosity of the product increases exponentially.2 From a formulation point of view, higher viscosities could cause problems with the drug’s syringeability (i.e. the ease with which the formulation can be pushed through a needle).3 At the patient level, the force and needle bore size needed to administer a viscous injection may result in increased discomfort and user anxiety.
THE SHL APPROACH
Benefitting from two decades of experience, SHL’s technologies are able to deliver any formulation for subcutaneous and intramuscular injections effectively and efficiently. We believe that an advanced drug delivery solution can save time and money in drug formulation without compromising patient comfort.
SHL’s offerings are patient-friendly autoinjector solutions for many different types of formulations, including:
- High volume
- High viscosity
- Unstable formulations requiring separation and reconstitution of drug and diluent.
Our most requested specifications are available in preconfigured devices that offer a faster developmental timeline compared with bespoke products, giving clients a much shorter path to market with their innovative and novel therapies.
For higher viscosity drug preparations, SHL’s Bertha® (Figure 1) provides the safe and reliable delivery of up to 60 centipoise. Bertha® is a disposable, fixed-dose autoinjector that is compatible with either a 1.0 mL or 2.25 mL prefilled syringe.

Figure 1: Bertha® is a disposable autoinjector that can safely deliver drugs with viscosities of up to 60 centipoise.
Made for higher viscosities, Bertha® was designed with a particular focus on human factors, as complex formulations can be harder for a patient to inject independently. Bertha® features a continuous clicking mechanism, visual feedback ribbon and viewing window for easily understandable and immediate feedback. Its larger design provides the user with a sense of security during the injection process, and its proprietary rounded needle cover helps to prevent accidental activation when the user is seeking the desired injection site.
For viscosities of up to hundreds of centipoise, SHL’s market-proven Rotaject® (Figure 2) technology offers safe and dependable support. Rotaject® can solve some of biologics’ most daunting challenges with its clock-spring technology, ensuring that the full dose is pushed by a constant force. This technology allows for high viscosity drugs to be delivered safely and comfortably, within seconds.

Figure 2: The Rotaject® technology uses a constant force technology that allows the delivery of highly viscous formulations of up to several hundred centipoise.
For dosing in higher volumes, SHL’s Molly® family of autoinjectors (Figure 3) can deliver up to 2.25 mL. Molly® 2.25, the highest volume variant, features the same simple two-step handling, ergonomic cap and automatic locking needle cover features of its predecessors. Its enhanced anti-roll feature prevents unwanted rolling for enhanced safety.

Figure 3: The Molly® family of devices (from left to right): Molly 1.0 mL FNS, Molly 1.0 mL RNS and Molly 2.25 mL.
SHL’s cartridge-based solutions, with a maximum fill volume of 3.0 mL, can safely deliver drugs in higher volumes. The Maggie®, for example, uses a 3.0 mL standard ISO cartridge. The VSDI®+NIT® autoinjector, compatible with 1.5 mL to 3 mL cartridges, allows precise dosing variations, customised to client needs.
“Benefitting from two decades of experience, SHL’s technologies are able to deliver any formulation for subcutaneous and intramuscular injections effectively and efficiently…”
Cartridges offer a broad range of options for fulfilling various drug characteristics and therapeutic needs, widening the scope of container choices, including for both single- and dual-chamber therapy solutions. However, because their needles are not pre-attached, they do pose a challenge in terms of avoiding contamination and preventing needlestick injuries. SHL addresses this problem with Needle Isolation Technology (NIT®), a unique safety solution where the needle is pre-installed in the device. In one simple step, the user initiates the automatic needle attachment process without being exposed to the needle. With this technology, we ensure that the needle is permanently hidden throughout the entire process, and we mitigate the risk of metal contamination.
For the drugs we handle, and for sensitive biologics in particular, SHL evaluates the leachability of syringes and primary containers on a case by case basis, in order to establish the optimal packaging for ensuring product stability.
Meanwhile, all of SHL’s offerings are developed in close partnership with biopharmaceutical clients to increase patient acceptance and safety throughout the injection process. Our in-house design team works with clients to develop devices based on both human factors studies and input from a range of stakeholders and experts on how the patient understands the device.
CONNECTED THERAPEUTICS
In recent years, SHL Group has integrated connectivity with its devices to enhance patient adherence, one of the most important factors affecting therapeutic outcome. Combining our own expertise with that of active partnerships, SHL is working on a number of initiatives in the digital space to bring innovation to the healthcare market.
Last year, SHL Group entered into a strategic partnership with QuiO, a New York-based digital healthcare start-up, to support even the most viscous and/or high volume biologics. QuiO is developing a range of smart add-ons for SHL’s devices, including SHL’s solutions for drug formulations with higher viscosities or volumes. QuiO is also developing a universal, reusable smart injector that will be compatible across all common primary containers, enabling drugs along a sweeping range of viscosity levels to be administered subcutaneously.

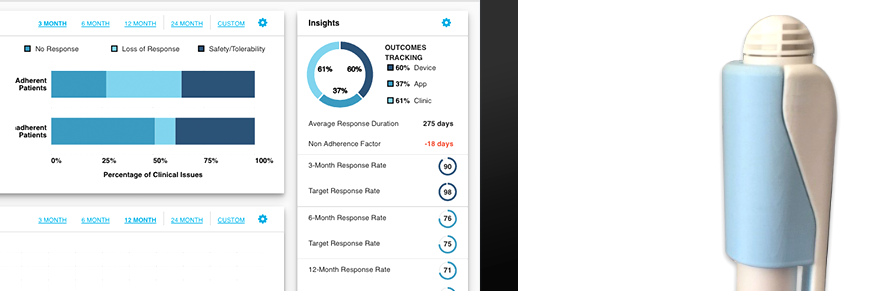
Figure 4: ConnectedRx® is the first solution designed specifically for connected therapeutics.
QuiO-connected devices can record injection performance and transmit that information to ConnectedRx®, a fully integrated cloud-based platform that is the first solution designed specifically for connected therapeutics (Figure 4).4 ConnectedRx® gives patients analytics and communication tools so that they can monitor their health metrics, and it can be paired with services like QuiO Coach™, which provides high-touch interventions and patient onboarding.
ConnectedRx® can also automatically collect dose-level data and share it as desired with stakeholders, including physicians and payers. Through the software, clinicians can track patient data indicating health status and medication adherence outside the clinic; they can also receive alerts for identifying patients in need of attention and are provided with tools for co-ordinating, performing and tracking interventions.
On the platform, payers and pharmaceutical clients can gain invaluable business intelligence derived from de-identified data measuring patient experience, adherence and health outcomes. These analytics can help them identify opportunities to improve adherence and to participate confidently in value-based contracts.
Integrated with ConnectedRx®, SHL’s injectors offer the unique opportunity of time-tested yet leading-edge delivery solutions that factor in the complexities of higher volume and higher viscosity drug preparations. Amid rapid growth in the biologics space, SHL’s delivery technologies enhance product differentiation for clients, as they support the realisation of protein-based therapies by making at-home administration comfortable, simple and safe, ultimately leading to better clinical outcomes.
REFERENCES
- Otto R, Santagostino A, Schrader U, “Rapid growth in biopharma: challenges and opportunities”. McKinsey & Company Insights, Dec 2014.
- Palm T et al, “The Importance of the Concentration-Temperature- Viscosity Relationship for the Development of Biologics”. BioProcess International, Mar 2015.
- Adler M, “Challenges in the Development of Pre-filled Syringes for Biologics from a Formulation Scientist’s Point of View”. Am Pharm Rev, Feb 2012.
- Dahmani A, “Software and Services – The Missing Link for Connected Therapeutics”. ONdrugDelivery Magazine, Issue 81 (Dec 2017), pp 10-14.