Citation: Shue S, “SMPFS: a Novel Prefillable Syringe with a Glass Primary Container but No Need for Extreme Thermal Reforming” ONdrugDelivery Magazine, Issue 107 (May 2020), pp 56-57.
Stephen Shue introduces a proprietary prefillable syringe, the SMPFS, whose novel glass-tube-plus-plastic-sheath design enables the use of a standard glass tube primary drug container, but avoids the need for the extreme thermal reforming process that conventional glass prefillable syringes must undergo.
The global market for prefilled syringes (PFS) was valued at US$4.9 billion (£3.9 billion) in 2018 and is expected to exceed $9.7 billion by 2025, according to Global Markets Insights.
“The use of standard glass tubing, simply cut to size but with no thermal reforming, means that E&L can be dramatically reduced.”
The increasing prevalence of chronic diseases such as diabetes and rheumatoid arthritis – which need prolonged drug administration in accurate doses – is one of the reasons behind the growth. PFS can be used to enhance patient compliance and dose accuracy. The emergence of biologics, which often need to be injected, has been another key driver. As well as ease and accuracy of use, benefits of PFS include increased product lifespan, minimised drug wastage – and elimination of the task of transferring a drug from a vial to a syringe, along with a corresponding decrease in the risk of drug contamination.
More than two billion PFS are used worldwide each year – with at least 60 drugs and vaccines available in a prefilled format for use across more than a dozen therapeutic categories. With analysts predicting that, by 2024, sales will reach around 12.4 billion PFS each year, there’s never been a better time to focus on producing a world-class PFS.
STANDARD GLASS PFS DRAWBACKS
Almost all the drawbacks of the conventional glass PFS originate from the extreme thermal reshaping process that they must go through, which reforms a segment of standard glass tubing into a glass syringe. The drawbacks include:
- Higher manufacturing cost
- Destabilised stored drug due to drug-container interactions induced by fume deposits
- Lamellae, leachables and extractables associated with reshaped glass syringes
- Fragility issues arising from reshaped portion of glass.
SaferMed has designed the SaferMed Prefillable Syringe (SMPFS), a novel proprietary PFS design comprising two main parts:
- A primary drug container consisting of a glass tubing segment made from the direct cutting of original long glass tube, in combination with suitable elastomeric components
- A plastic syringe, made using standard precision mould injection mass production processes, which functions as a protective sheath fixed around the glass tube.
Together these components form the function orientated PFS. SaferMed has focused on eliminating the drawbacks of conventional glass PFS, which must be shaped from a segment of glass tubing heated to extreme temperatures.
“The SMPFS, covered by several US and Taiwan patents, with patent applications pending in other areas, is currently at the concept stage and SaferMed Technologies seeks partners to advance its development.”
With the SMPFS, there is no need for the extreme thermal reshaping processes to be applied to the glass tubing drug primary container – and so no need for complex manufacturing facilities. All components are easily sourced from customised suppliers.
Easier and more efficient manufacturing means a lower overall cost for SMPFS – as low as one-third to half of the cost of conventional PFSs. The list of benefits also includes a reduction in the energy used during the glass manufacturing process.
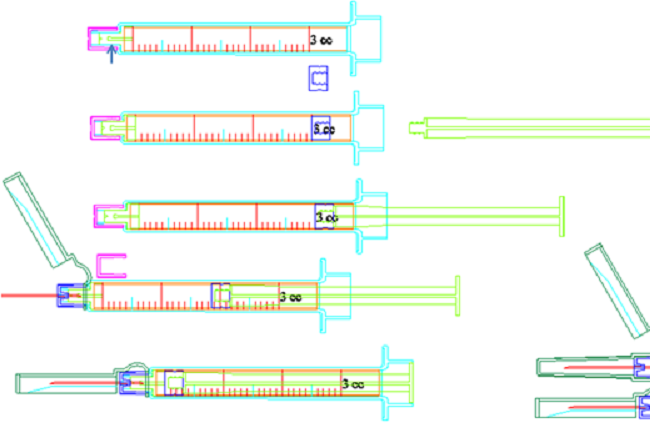
The integrated plastic sheath protection of the SMPFS solves the fragility issues of conventional glass PFSs – providing safer transportation and stockpiling. The plastic component also provides the familiar syringe form and function (syringe barrel, finger flange and tip) required for assembling the additional components such as the plunger and needle hub with needle safety shield (see Figure 1).

Figure 1: Main components of SaferMed’s SMPFS (shown here is a 3 mL variant with detachable needle and front elastomeric stopper).
The use of standard glass tubing, simply cut to size but with no thermal reforming, means that extractables and leachables (E&L) can be dramatically reduced or even eliminated, especially metallic ions and salts extracted by drugs, fume deposits, lamellae, and residual tungsten and glue often found in conventional glass PFSs. This confers the additional advantage of longer shelf life, including for vaccines, due to minimised or eliminated contaminants that are commonly found in traditional glass PFSs. SaferMed’s innovative technologies can be applied to produce devices in various volumes and configurations, all based on the current state-of-the-art technologies that provide physically and chemically stable elastomers, and within the paradigms of currently marketed glass PFS and cartridges with elastomeric components in situ. For example, SMPFS can be configured with a detachable needle with front elastomeric stopper, and with a permanent needle, and in 1 mL and 3 mL volumes. Large-volume PFS for the delivery of antibiotics, and devices with volumes up to 25 mL are also possible.
More than two billion PFSs are used worldwide each year – with at least 60 drugs and vaccines available in a prefilled format for use across more than a dozen therapeutic categories. With analysts predicting that, by 2024, sales will reach around 12.4 billion PFSs each year, there’s never been a better time to focus on producing an innovative, improved yet lower-cost PFS.
BUSINESS STRATEGY
The SMPFS, covered by US and Taiwan patents, with patent applications pending in other areas, is currently at the concept stage and SaferMed Technologies seeks partners to advance its development – an investor or device partner, to develop the device further, or a pharma partner to develop a pharmaceutical or biologic product that uses the devices.
In addition to the benefits around reduced E&L, reduced fragility, increased stability, longer shelf life, and reduced cost of goods described above, with its SMPFS SaferMed aims to reduce needle-stick injuries to needle-based delivery device users, to make the use of PFS easier and more convenient for patients and healthcare workers, and to enable a syringe application that is more environmentally friendly.
The ultimate objective is, in collaboration with a partner, to introduce this beneficial technology to the biopharmaceutical industry.