Citation: Morin C, “Synergising Excipients to Boost Skin Delivery”. ONdrugDelivery Magazine, Issue 99 (Aug 2019), pp 54-58.
Cécile Morin and Delphine Marchaud discuss combining polar and apolar excipients in topical formulations in order produce synergies that maximise the performance of the drug.
The skin constitutes a natural barrier to prevent loss of water from the body and penetration of exogenous substances into it. Understanding its constitution and organisation is essential when formulating efficient topical or transdermal dosage forms.
The “brick and mortar” representation is commonly used to describe the stratum corneum (SC), with the corneocytes being the “bricks” and lipids the intercellular “mortar”. A gradient of decreasing lipophilicity is observed from the upper layer of the SC down to the dermis.
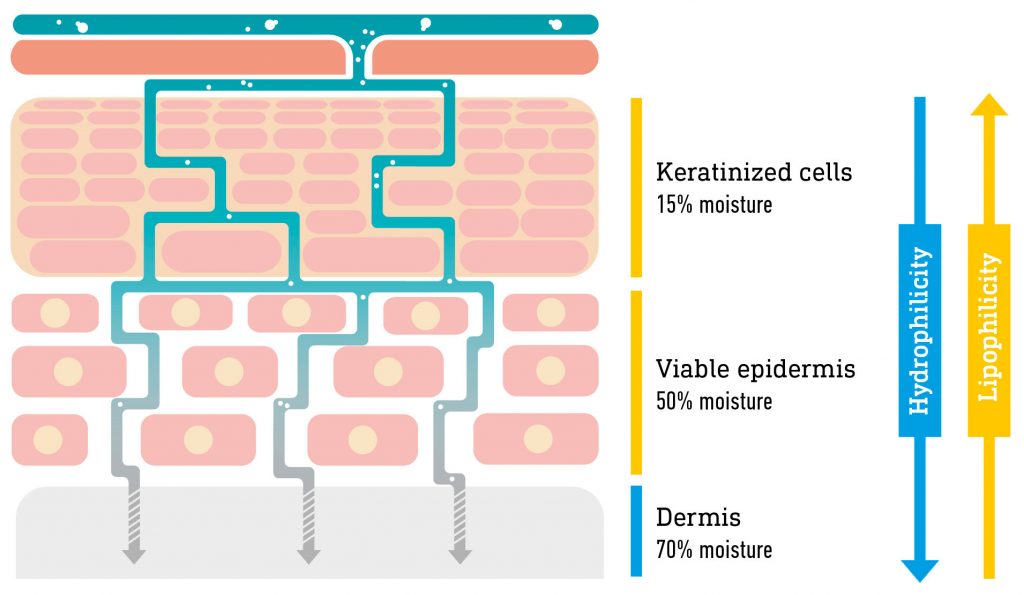
Figure 1: Schematic representation of the structure of the skin.
How vehicles in a formulation interact with the lipid structure significantly contributes to drug diffusion through the different layers of the skin. The major route of drug diffusion through the epidermis is the intercellular path1 (Figure 1).
Solubility: the formulation must solubilise a sufficient amount of drug to reach an effective concentration at the target site.
Three main steps govern drug diffusion from the formulation to the skin:
- Partition: the drug must partition out of the delivery vehicle into the upper layers of the SC.
- Diffusion: the drug molecule diffuses through the SC mainly via the intercellular path.
Therefore, the art of formulation consists of choosing the appropriate vehicles and determining their correct ratio to maximise drug solubility in the formulation and subsequent partition in the skin.Fick’s law applies for passive diffusion, meaning it is driven by drug concentration and that maximum thermodynamic force is obtained at saturation solubility.
Gattefossé has a wide range of lipid excipients for skin delivery, some of which are described in Table 1. This article will focus on Transcutol® P, Capryol™ and Lauroglycol™ and the benefits that arise from their synergistic combination in topical formulations.
| Trade Name | Chemical Name | Practical Hydrophile-Lipophile Balance (HLB) |
| Capryol™ PGMC | Propylene glycol monocaprylate (type I, monoesters >55%) NF |
6 |
| Capryol™ 90 | Propylene glycol monocaprylate (type II, monoesters >90%) NF |
5 |
| Lauroglycol™ FCC | Propylene glycol monolaurate (type I, monoesters >45%) EP/NF |
5 |
| Lauroglycol™ 90 | Propylene glycol monolaurate (type II, monoesters >90%) EP/NF |
3 |
| Transcutol® P | Highly purified diethylene glycol monoethyl ether EP/NF |
N/A |
Table 1: Gattefossé’s main excipients for dermal drug delivery (NF – Compliant with National Formulary monograph, EP – Compliant with European Pharmacopoeia).
LIPID EXCIPIENTS: NATURALLY ADAPTED TO THE SKIN
Lipid excipients, with their high solubilising power and amphiphilic properties, enable modulation of the penetration of the active pharmaceutical ingredient (API) into the SC, and drive API flux.
Transcutol® P is a safe and effective hydrophilic solvent widely used in skin delivery.2 Transcutol® increases the solubility of both lipophilic and hydrophilic APIs. Furthermore, it can penetrate the SC and interact with the water in the intercellular space.3
Capryol™ and Lauroglycol™ are lipophilic solubilisers. They consist of fatty acid esters and can interact with the lipids in the intercellular space. The greatest permeation is observed with excipients containing caprylate (C10) and laurate (C12) fatty acid esters, whereas myristate (C14) and stearate (C18) favour skin-vehicle partitioning.4
Polar solvents (e.g. Transcutol®) increase drug solubility in the SC, whereas non-polar solvents (e.g. Capryol™, Lauroglycol™) increase the diffusion parameter of the drug in the SC. Their combination in a formulation has been reported to deliver higher efficiency.
A PROVEN SYNERGISTIC EFFECT
The combination of solubilisers is a common practice to maximise drug solubility, thermodynamic force and partition in the skin. Combinations of Transcutol® with other permeation enhancers have been reported in the scientific literature and reviewed by Osborne & Musakhanian (2018). Examples of synergistic combinations using Transcutol® and either Capryol™ or Lauroglycol™ are detailed hereafter.
Example One: Neat Solvents – Ketorolac Tromethamine – Rodent Skin
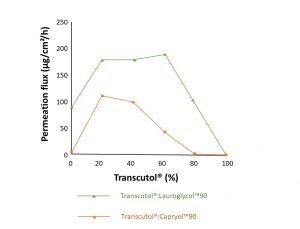
Figure 2: Ketorolac tromethamine flux in neat solvent and solvent mixtures (Adapted from Cho et al, 2004).
Cho et al (2004)5 studied the transdermal delivery of ketorolac tromethamine. The study measured drug solubility and permeation through excised hairless mouse skin in various excipients, pure or in mixtures.
Permeation flux was highest for Lauroglycol™ and solubility highest for Transcutol® (Table 2). They then combined Transcutol® with either Lauroglycol™ or Capryol™ at different ratios (Figure 2). A 20% increase in permeability was observed for combined Transcutol® and Capryol™ in the ratios 80:20 and 40:60. A two-fold increase in permeation over Lauroglycol™ alone was observed for combined Transcutol®:Lauroglycol™ mixtures in the ratios 20:80, 40:60 and 50:50.
To the authors’ knowledge, this study was the first to report a synergistic relationship between the polar solvent Transcutol® and apolar solvents such as Capryol™ and Lauroglycol™.
| Solubiliser | Permeation flux (μg/cm²/h) | Solubility (mg/mL) |
| Capryol™ 90 | 6.08 ±2.29 | 51.3 ±5.68 |
| Lauroglycol™ 90 | 94.3 ±17.3 | 15.2 ±1.87 |
| Transcutol® P | 0.69 ±0.29 | 211 ±11 |
Table 2: Flux and solubility of ketorolac tromethamine with different solubilisers.5
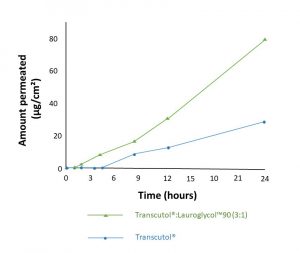
Figure 3: Genistein permeation from a gel containing 25% permeation enhancers (Adapted from Chadha et al, 2011).
Chadha et al (2011)6 formulated a gel with genistein and various permeation enhancers and assessed permeation across human skin (Figure 3). A three-fold increase in genistein solubility was observed with Transcutol® alone, whereas the combination Transcutol®:Lauroglycol™ 90 exhibited a 12-fold increase versus the control formulation with ethanol. Similarly, a significant five-fold increase in flux was achieved with Transcutol® alone and a 13-fold increase when combining Transcutol® and Lauroglycol™ 90 (ratio 3:1).
Example Two: Gel Formulation – Genistein – Human Skin
This study confirmed that the synergistic combination of Transcutol® and Lauroglycol™ is efficient when formulated in a gel and tested on human skin.
Example Three: Neat Solvents – Carbenoxolone – Human Skin
Hirata et al (2013)7 tested various solvents, pure or in association, and assessed the skin permeability of carbenoxolone on human skin (Figure 4).
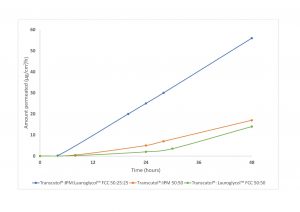
Figure 4: Carbenoxolone permeation from binary and ternary solvent mixtures (Adapted from Hirata et al, 2013).
Dimethyl isosorbide (DMI), isopropyl myristate (IPM) and Transcutol® were tested as neat solvent, but permeation at 24 hours was below 0.3 μg/cm². In binary mixtures, synergies were observed, and permeation reached 16.0 and 14.0 μg/cm² at 48 hours for the binary systems Transcutol®:IPM and Transcutol®:Lauroglycol™ FCC, respectively.
Although binary mixtures of solubilisers significantly improved the permeation, the maximum synergy was observed with the ternary composition consisting of Transcutol®:IPM:Lauroglycol™ FCC in the ratio 50:25:25, with flux reaching 56 μg/cm²/h.
This study highlights that for some actives, a ternary solubiliser system is needed to reach sufficient level of permeation.
Example 4: Gel Formulation – Diclofenac Sodium – Human Skin
At Gattefossé, diclofenac sodium was used as a model drug and formulated in gels for permeability studies on human skin (Figure 5). Although the permeation obtained with the binary system propylene glycol (PG):Lauroglycol™ FCC was quite high (about 100 μg/cm²), the best performance was achieved with a quaternary system consisting of Transcutol®P, IPM, PG and Lauroglycol™ FCC (Figure 5) and a permeation of about 120 μg/cm².
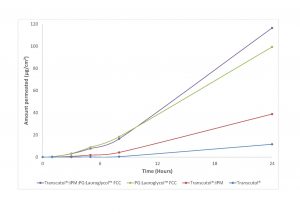
Figure 5: Diclofenac sodium permeation through human skin from various gel formulations (Gattefossé in-house study).
This study highlights the fact that some drugs are best served by multi-component solubiliser systems, with three to four excipients, to maximise drug delivery through the skin. The formulator has to determine the optimal ratio of polar and apolar solvents for the drug, and this can only be done on a case-by-case basis.
CONCLUSION
Drug delivery to the skin is a challenging process requiring solubilisation, partition and diffusion of the drug through the different dermal layers. Each drug is specific and no general rule can be established. Therefore, a case-by-case approach is required to determine which solvents (polar and apolar) are best suited to the drug. However, when developing a formulation, one has to keep in mind that even when permeation is low with individual excipients, combination can produce synergies, as was demonstrated with Transcutol® P in association with Lauroglycol™ or Capryol™.
REFERENCES
- Lane ME, “Skin penetration enhancers”. Int J Pharm, 2013, Vol 447(1–2), pp 12–21.
- “Efficient skin delivery: no compromise with Transcutol®”. Gattefossé Whitepaper, 2015.
- Osborne DW, Musakhanian J, “Skin Penetration and Permeation Properties of Transcutol® – Neat or Diluted Mixtures”. AAPS PharmSciTech, 2018, Vol 19(8), pp 3512–3533.
- Aungst BJ, Rogers NJ, Shefter E, “Enhancement of naloxone penetration through human skin in vitro using fatty acids, fatty alcohols, surfactants, sulfoxides and amides”. Int J Pharm, 1986, Vol 33(1–3),
pp 225–234. - Cho YA, Gwak HS (2004). “Transdermal delivery of ketorolac tromethamine: effects of vehicles and penetration enhancers”. Drug Dev Ind Pharm, 2004, Vol 30(6), pp 557–564.
- Chadha G et al, “In vitro percutaneous absorption of genistein from topical gels through human skin”. Drug Dev Ind Pharm, 2011, Vol 37(5), pp 498–505.
- Hirata K et al, “Formulation of carbenoxolone for delivery to the skin”. Int J Pharm, 2013, Vol 448(2), pp 360–365.





