Citation: Joughin J, “Biologic Drugs and their Delivery Systems: A Symbiosis Key to Commercial Success”. ONdrugDelivery Magazine, Issue 90 (Sep 2018), pp 10-13.
Jeannie Joughin discusses the ever-growing importance of the delivery system in the biologic combination product area, highlighting how it is the delivery system, not the drug, that defines the patient experience.
“The distinctions between a drug and its delivery system have been falling away for years, just as predicted by Deloitte…”
There’s a new wind in the healthcare industry. Pharma industry meetings are focusing heavily on it. Regulatory authorities are establishing new departments and guidelines to review and approve it. Companies developing biologic drugs are streaming in for site visits and signing development deals to attain it. Patients in focus groups and participants in human factors trials say they prefer it. What is this new wind that pharma companies, regulatory authorities, patients and healthcare providers are turning their focus to? It’s the “combination product”, specifically the full integration of biologic drugs with their delivery system in the form of wearable injectors. These biologic-delivery combination products are becoming mainstream, and the addition of higher delivery volume combination products will greatly benefit users, as well as help to control healthcare costs.
But that is not what the paradigm shift is about. Rather, it’s the realisation that biologic drugs and their delivery mechanism share a symbiotic relationship as closely aligned as the one between a bow and arrow. There is little value in one without the other.
SYMBIOSIS: BOW AND ARROW
The distinctions between a drug and its delivery system have been falling away for years, just as predicted by Deloitte.1 As the life sciences consultancy discerned some time ago, the integration of technology and pharma products has indeed gained traction and accelerated. Why? Because the drug itself represents a diminishing portion of the whole that comes together to deliver an overall outcome. Outcome is the key metric for today’s healthcare systems and is increasingly being assessed with big data and artificial intelligence (AI).
To achieve the desired outcomes, patients first have to take the drugs. Therefore, what’s good for patients – easy, painless, less costly, more convenient biologic drug administration – is very good for business. Take Amgen’s Neulasta® (pegfilgrastim): despite its loss of patent protection in 2015, the addition of an on-body injector has been effective at extending Neulasta’s lifecycle. Because Neulasta must be given the day after chemotherapy, the wearable injector allows patients to administer the dose at home without having to return to a healthcare provider. Tony Hooper, Amgen’s Executive Vice-President of Global Commercial Operations, told investors during a recent call that, with regard to Neulasta, “We continue to drive increasing adoption and [the combination product] now represents over 50% share of all Neulasta purchases. This has been a great example of a very successful lifecycle management strategy.”2
A WINNING PATIENT ENGAGEMENT STRATEGY
Amgen has found a key to patient engagement with a winning combination product. Without the integration of their drug with a delivery system that patients found far more convenient, would Neulasta be as commercially successful in the face of biosimilar approvals? It’s doubtful.
“For preloaded injectors needing refrigeration, the patient has to wait for 30 minutes or more for the drug/device to warm to room temperature…”
Other pharma executives are thinking about patient engagement as well, more than the general news or trade press would have one believe. For example, most interviews with GSK’s new head of R&D, Hal Barron, result primarily in reports about the drug pipeline and its implications for the company’s business. But apparently there’s more on his mind. In a television interview on business channel CNBC, discussing GSK’s investment in 23andMe, Barron had the opportunity to slip in that, in addition to obtaining access to 23andMe’s vast trove of data, GSK would have the benefit of access to engaged patients, those who have given permission for their genetic information to be used in research. That cohort of accessible patients could be invaluable to GSK.
“Patient value has now become synonymous with business value,” said Josh Bramwell, Director of industry group eyeforpharma. “Companies listening to the growing patient voice and incorporating those insights into the design of products, trials and strategy are being rewarded with higher uptake and better outcomes.”
DELIVERY, NOT BIOLOGIC, DEFINES THE PATIENT EXPERIENCE
From the patient perspective, the delivery device is the element that defines the treatment experience more than any other. Delivery is especially challenging for large-volume biologic drugs, which in most cases are still being administered intravenously (IV). In addition to adding significant costs, IV administration is unpopular with patients. It is inconvenient. It can be painful. It is not a promising path for pharmaceutical companies racing to improve outcomes and be recognised as patient centric.
Subcutaneous injection is faster and more convenient, thus tends to be preferred by patients over IV. A time-and-motion study undertaken in eight countries reported significant time-savings for both healthcare professionals and patients through use of subcutaneous rituximab versus IV rituximab. The findings suggest potential for reduced waiting times, greater appointment availability and improved efficiency of oncology units with the subcutaneous formulation. Compared with IV drugs, the majority of participants in the study considered subcutaneous drugs clinically safer and more cost-effective, resulting in higher patient satisfaction.3
And now, as with Neulasta, subcutaneous injections can be administered by the patient at home or work by employing new, user-friendly delivery technology. Such injections are not generally painful and carry a reduced risk of infection, needlestick injuries and other complications. These wearable large volume injectors, with no needle in sight, make it even easier and less fearful for patients to administer their own treatment. It’s of benefit not only to patients but to the caregivers tasked with giving injections.
Indeed, Accenture Life Sciences found that 69% of patients said product benefits were a top factor in their treatment decisions and suggested that pharma companies should be thinking about promoting benefits that appeal to patients. Many patients would welcome an alternative to an IV infusion that doesn’t require a time-consuming, inconvenient trip to a health facility. It could burnish the brand, just as Neulasta’s combination product has done for Amgen.
ADDITIONAL PATIENT PREFERENCES TO CONSIDER FOR SPEEDIER ADOPTION
Patient concerns are another reason for the must-have partnership between biologic drugs and their delivery device. There are questions delivery device companies ask that pharmaceutical companies may not, questions relevant to actualising the patient-centric approach that speeds patient adoption.

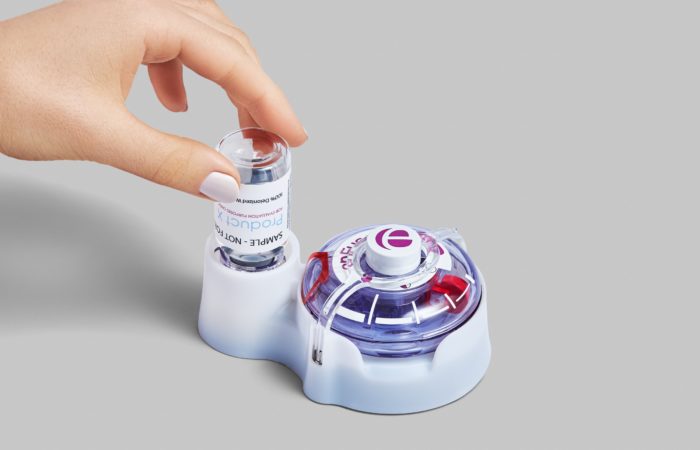
Figure 1: The EnFuse™ patient-loaded wearable injector.
For example, across dozens of human factors studies, Enable Injections has found that, surprisingly, patients receiving high volume (up to 50 mL) preferred patient-loaded to pre-loaded devices. It’s counterintuitive. Almost anyone’s initial reaction when presented with the choice “preloaded” or “patient-loaded” would be to assume that a preloaded device is preferable. In the case of non-refrigerated, small-volume (<5 mL) drugs, they may be right. But in the case of a large-volume, refrigerated drug, most patients preferred the Enable Injections enFuse™ patient-loaded system (Figure 1). Why? They no longer had to wait. For preloaded injectors needing refrigeration, the patient has to wait for a long period, typically 30 minutes or more, for the drug/device to warm to room temperature. At colder temperatures the drug viscosity increases significantly, causing a painful injection.
When using the enFuse device, the patient stores the vial or syringe in the refrigerator but not the delivery mechanism. When ready, the patient removes the container from the fridge and inserts it into the enFuse system, which automatically and passively warms the drug during the transfer to the injector. The injector is ready to use immediately after it is filled, a total wait of about a minute or so.
Other reasons patients gave for their preference for patient-filled devices were:

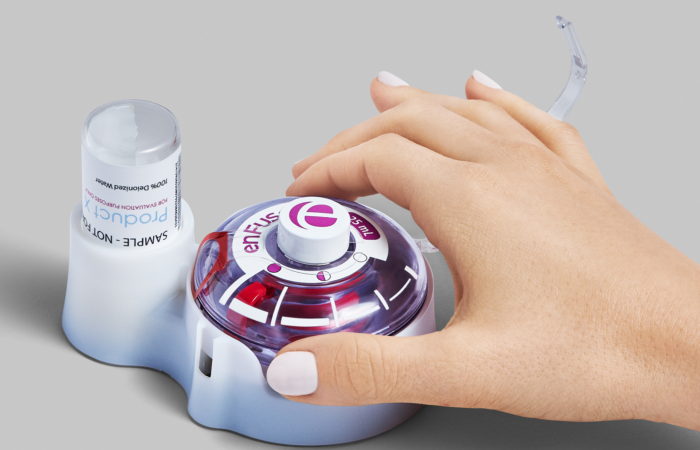
Figure 2: The Smart enFuse™ companion app.
- The vial doesn’t take up much room in the refrigerator.
- The vial is childproof, an injector isn’t.
- Once I start something I want to finish it.
- If I have to leave it out I might forget about it.
Since patient acceptance is a top priority, not only are needles never visible, but the most advanced on-body delivery systems (OBDS) also incorporate pause control for optimum personalised comfort, and connectivity to monitor adherence (Figure 2). They can deliver doses as large as 50 mL. Their small profile allows them to be easily concealed under clothing, leaving patients free to ambulate and go about their daily routine during treatment (Figure 3). There are other things on patients’ minds as well. “The opportunities in wearable devices are vast,” says Ashley Whitney at Cambridge Consultants. “As drugs evolve to improve patient care, however, device designs must evolve in parallel. Patients with chronic diseases do not want to be defined by their disease; wearable devices offer a brief reprieve from the reminder that they are living with an illness.”4

Figure 3: enFuse™ has a small profile, minimising its intrusion into patients’ daily lives.
FEEDBACK FROM CLINICAL TRIAL PATIENTS
“There are questions delivery device companies ask that pharmaceutical companies may not, and they are relevant to actualising the patient-centric approach that speeds patient adoption…”
Finally, the importance of the patient perspective extends to today’s clinical trials, where the patient’s role is pivotal. It has become much more important in trials to have a representative person experience and comment on the treatment. Physiological, safety and efficacy data are of course gathered, but patients are now reporting back on how the treatment experience feels, how it could be made easier, what they liked and disliked about it. In pharmaceutical company patient panels, Enable Injections’ enFuse technology was preferred to other delivery methods, providing some assurance that patients would report a positive experience.
EVEN THE FDA IS “LEANING IN” ON COMBINATION PRODUCTS
Gone are the days of the US FDA approving drugs based on their clinical trial data alone. The regulatory agency has established an Office of Combination Products to meet the demand for advanced treatments that combine drugs and delivery.5 Can there be any further doubt that in categories in which existing injection technology is inadequate for administration, biologic drugs and their delivery technology are, like a bow and arrow, essentially inseparable?
REFERENCES
- “2016 Global Life Sciences Outlook”. Deloitte, Report, 2015.
- Bulik BS, “Amgen adds online cancer team narratives to Neulasta Onpro push”. FiercePharma, May 2017.
- Doughty D et al, “Understanding subcutaneous tissue pressure for engineering injection devices for large-volume protein delivery”. J Pharm Sci, Jul 2016, Vol 105(7), 2105–2113.
- Whitney A, “Drug Delivery On The Go: Device Design Considerations”. Med Device Online, Nov 2015.
- Hunter NL, Sherman RE, “‘Leaning in’ on Combination Products”. FDA Voice, March 2016.