Citation: King J, “Drug Delivery Designed to Improve the Patient Experience”. ONdrugDelivery Magazine, Issue 105 (Feb 2020), pp 52-55.
Jennifer King looks at the delivery challenges of novel therapies for chronic diseases using a specific patient’s story as an example, going on to highlight the ways in which a novel device approach could make all the difference for patients.

Figure 1: The enFuse, on-body infusor.
Chronic illness touches everyone, it seems. Whether it is a family member, friend or co-worker, many people have some understanding of how difficult life can be while managing a chronic disease. The biotech revolution has led to many novel therapies that are changing lives. But these new therapies may come with delivery challenges that make it difficult for a patient to receive treatment. When a patient shares stories of the rigours they endure to manage their disease, it highlights the motivating factor behind novel technology like the enFuse on-body infusor platform (Figure 1).
A REAL PATIENT WITH A REAL STORY
“In a survey of patients receiving IV enzyme replacement therapy, 40% felt that hospital-based ERT is disruptive, causing loss of days at school and work, stress and family issues.”
The Enable Injections team recently spent time with a patient who introduced an interesting perspective on her infusions. Megan has managed a chronic autoimmune disease for more than 15 years. She is passionate about helping others who live with similar autoimmune diseases – especially young people, because she was diagnosed at the age of 18.
Megan was initially prescribed 28 pills per day for treatment, which proved to be difficult to manage and ineffective. While planning her life around her dosing schedule, she lived in a mental fog due to fatigue and was unable to travel or do other activities she loves.
At the age of 23, she began seeing a new doctor who said to her: “You are a young person and you deserve to live your life.” Megan said hearing those words was life-changing, as she thought she would never feel well again. Her new doctor prescribed a new therapy via IV administration, and she began feeling better. Even though she was required to take an afternoon off work for each treatment, she didn’t mind the inconvenience. She began to think of the three hours tethered to the IV as “me time” because the hospital infusion centre was comfortable and, over time, she came to know the staff. However, each infusion had the potential for its own challenges.
For an IV infusion to go well, the medicine needed to arrive at the correct location at the correct time, the IV had to be placed at the site without complications, her vitals had to be in the right range and her body had to respond properly to the treatment. In addition, to receive her treatments, Megan often had to work with her insurance company to gain the proper approvals to ensure her treatments were covered.
Recently, Megan’s insurance payer interrupted her IV infusion routine by informing her that it now requires home infusion for her IV therapy. In Megan’s words, setting up the home infusion was a disaster. The home infusion nurse showed up hours late, the supplies and medicine arrived a day late and, when they did arrive, she had to spend three hours sitting on the couch with a stranger in her home. This was uncomfortable for Megan because she lives alone.
Megan asked the infusion nurse what she would do if there was a reaction. The nurse responded that she wasn’t sure and mentioned calling the 911 emergency number. Megan felt uncomfortable with the arrangement to get emergency care in the event of a reaction.
As a result of her concerns, Megan’s insurance now allows her to receive her IV infusions at the home infusion company site. However, she receives her infusion in a windowless cinder block room, and still faces the same logistical challenges, such as missing time at work, as she did when she was treated in a hospital. With no alternative treatment options, she feels stuck and lacking control over her treatments.
CURRENT INFUSION OPTIONS AND CHALLENGES
From the patient’s perspective, the potential move of infusions from healthcare clinic to provider-administered at home has many challenges. In a survey of patients receiving IV enzyme replacement therapy (ERT), 40% felt that hospital-based ERT is disruptive, causing loss of days at school and work, stress and family issues. However, 93% of patients had the perception that receiving ERT in the hospital had the advantage of greater safety, closer monitoring and more support from health professionals compared with at-home treatment. A total of 55% were willing to receive ERT at home but 33% were against it.1
While home infusion options bring benefits to the payer insurance companies through savings in overall treatment costs2, as reimbursement changes, patients may experience an increased burden from medical versus pharmacy benefits. These challenges may force other choices onto the patient – including, ultimately, whether to continue care. In the end, for real people with conditions that require energy to manage, the patient usually needs to remain on their therapy to experience the benefits that come with improved health.
DESIGNING TO ALLEVIATE CHALLENGES
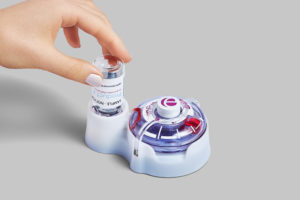
Figure 2: The enFuse 25 mL on-body vial transfer system.
Enable Injections has focused on the patient right from the start – it was originally founded to help improve the way children receive vaccinations, in conjunction with research being conducted at Cincinnati Children’s Hospital (OH, USA). Soon after work began, Enable’s key objective shifted to focus on an even more pressing matter – a way to help patients receive their injectable medicines outside of a healthcare facility. To support the emergence and growing importance of biologics therapies, which often require administration in large volumes, the enFuse technology was born.
“Patient preference studies indicate most patients prefer
home-based treatment over treatment at a healthcare facility.”
Through dozens of human factors studies, Enable Injections honed the enFuse design to simplify patient interaction with the device and target a large range of patient users. Using feedback from pharma and biotech companies, Enable Injections worked to incorporate one of the most pressing requests from pharma into the design – to use the original primary container closure to load the device for delivery for infusion via the enFuse system (Figure 2).
The enFuse platform is designed to leverage existing primary container closure systems, which eliminates the need for additional drug compatibility testing, additional filling and manufacturing lines, and additional stability testing risk. EnFuse is under development in 10, 25 and 50 mL variants (Figure 3).

Figure 3: The enFuse portfolio of on-body infusors is being developed in 10, 25, and 50 mL sizes.
BENEFITS OF SELF-ADMINISTRATION
Patient preference studies indicate most patients prefer home-based treatment over treatment at a healthcare facility.3,4 Homebased treatment is associated with fewer days of missed work and school compared with administration in a healthcare clinic.5, 6, 7 Patients report home-based treatments are more comfortable, less stressful, more effective and have less impact on family life.8
In a 12-month prospective evaluation of outcomes in patients with primary immunodeficiency, patients reported fewer limitations with work and daily activities, improved vitality and better general health after switching to subcutaneous home infusion from IV infusion. The majority reported they preferred the subcutaneous route of administration and home-based therapy over IV infusion.9
Studies also report economic benefits and cost reductions in home-based treatment versus hospital or clinic treatment. The cost savings have the potential to be realised from both a healthcare payer perspective and a patient and societal perspective.5
With self-administration, treatments may be more easily accessed by the patient. Patients may no longer be required to travel to a healthcare facility to receive treatment or schedule appointments with healthcare providers for in-home treatment. In addition, patients may be more likely to adhere to treatment regimens, as administration may not require additional time away from other activities.
Payers, policy makers, physicians and employers may appreciate the potential positive impact on the healthcare system. However, even with the benefits, the shift to home-based self-administration will require significant effort and collaboration between technology providers like Enable Injections, pharma innovators, payers, prescribers, pharmacies and providers to truly improve the patient experience.
THE GOAL OF MAKING A DIFFERENCE
At Enable Injections, our goal is to help make a difference for people. The enFuse is designed to help improve the patient experience. Enable Injections will continue to stay engaged with patients like Megan to help inform the development of the enFuse.
When asked about whether she would prefer her “me time” at the hospital sitting in the infusion clinic for hours or to regain time through the potential for home self-administered infusion, Megan said she would choose to have her time back. She wouldn’t have to stop working or travelling, invite a stranger into her home or arrange her life to accommodate infusion if she could self-administer at home. She would have more control over an aspect of her life that so often feels that it lacks control. For Megan, and other patients like her, that is what makes all the difference.
REFERENCES
- Parini R et al, “Intravenous enzyme replacement therapy: hospital vs home”. Br J Nurs, 2010, Vol 19, pp 892–894, pp 896–898.
- Perraudin C et al, “Switching Patients to Home-Based Subcutaneous Immunoglobulin: an Economic Evaluation of an Interprofessional Drug Therapy Management Program”. J Clin Immunol, 2016, Vol 36, pp 502–510.
- Smith A et al, “Home care versus hospital care in patients with multiple myeloma treated with pamidronate”. Int J Palliat Nurs, 2004, Vol 10, pp 144–149.
- Daly P, Evans J, Kobayashi R et al, “Home-based immunoglobulin infusion therapy: quality of life and patient health perceptions”. Ann Allergy, 1991, Vol 67, pp 504–510.
- Petraroli A, Squeglia V, Di Paola N et al, “Home Therapy with Plasma- Derived C1 Inhibitor: A Strategy to Improve Clinical Outcomes and Costs in Hereditary Angioedema”. Int Arch Allergy Immunol, 2015, Vol 166, pp 259–266.
- Vultaggio A et al, “Subcutaneous immunoglobulin replacement therapy in patients with primary immunodeficiency in routine clinical practice: the VISPO prospective multicenter study”. Clin Drug Investig, 2015, Vol 35, pp 179–185.
- Tourangeau L et al, “Safety and efficacy of physician-supervised selfmanaged C1 inhibitor replacement therapy”. Int Arch Allergy Immunol, 2012, Vol 157, pp 417–424.
- Milligan A, Hughes D, Goodwin S et al, “Intravenous enzyme replacement therapy: better in home or hospital?”. Br J Nurs, 2006, Vol 15, pp 330–333.
- Nicolay U et al, “Healthrelated quality of life and treatment satisfaction in North American patients with primary immunedeficiency diseases receiving subcutaneous IgG self-infusions at home”. J Clin Immunol, 2006, Vol 26, pp 65–72.

