Citation: Huddleston M, Eibling A, King J, “Key Challenges in Successful Wearable Drug Delivery & Patient Self-Administration”. ONdrugDelivery Magazine, Issue 100 (Sep 2019), pp 4-7.
Matthew Huddleston, Andrew Eibling and Jennifer King look at the key challenges involved in successful wearable drug delivery technology and patient self-administration.
Cutting-edge biologic therapies often require patients to receive treatment through frequent intravenous (IV) infusions at a hospital or infusion clinic. But what would happen if those same biologics could be given by the patient via a simple through-the-skin infusion at home? Why has there not been a greater push for wearable technology that allows people to self-administer infused medicines at home?
“There exists the need for a cost-effective, simple-to-use, large-volume, subcutaneous, self-administration infusion system which creates real value for both pharmaceutical companies and patients.”
Armed with game-changing technology which makes this a possibility, Enable Injections’ goal is to reduce the treatment burden on people living with a wide range of conditions and put control back into the hands of the patient.
The world is ready for the possibility. US Healthcare costs are increasing at a rate of 3-4% per year.1 Evidence shows a reduction in the overall cost when therapy is administered in the home compared with a doctor’s office or clinic.2 Patients are ready for it too. Evidence shows improved compliance and adherence to medications when patients can complete the therapy at home compared with administering infusion in the physician’s office or clinic.3
Up to this point, technology has been the limiting factor. Biologic therapies often start with IV infusion and require large volumes for efficacy. Transition to subcutaneous delivery may even require higher volumes due to bioavailability. These large volumes are not suitable for administration via autoinjectors and prefilled syringes. Large infusion pumps are available for subcutaneous delivery in the home but they are complex and often require in-home infusion services for administration.
There exists the need for a cost-effective, simple-to-use, large-volume, subcutaneous, self-administration infusion system which creates real value for both pharmaceutical companies and patients. With the right technology, the market environment is primed to transition.
PHARMA CHALLENGES
Cost
Patient-centric delivery devices must provide value and simplicity for the patient, whilst minimising development costs and maximising speed to market for the pharma company. The technologies being developed for the on-body infusor market are varied and introduce different development and manufacturing costs.
Container
Therapeutics start their lives in liquid or freeze-dried (lyophilised) form within standard vials. The challenge for large-volume, wearable delivery devices is that the standard container closures are not designed to be a component of a wearable delivery device. Autoinjectors (pens) and pumps have proven to be exceptionally valued for relatively small volumes (insulin) but the wearability of large volumes required for therapeutic dosing for biologics and biosimilars is a challenge.
To accommodate the challenge, large-volume infusor developers incorporate custom container cartridges into the delivery device design. The transition from a standard vial to a custom container introduces a host of other risks for pharma and biopharma companies, including extended stability testing, sterility issues, and packaging and transport challenges.
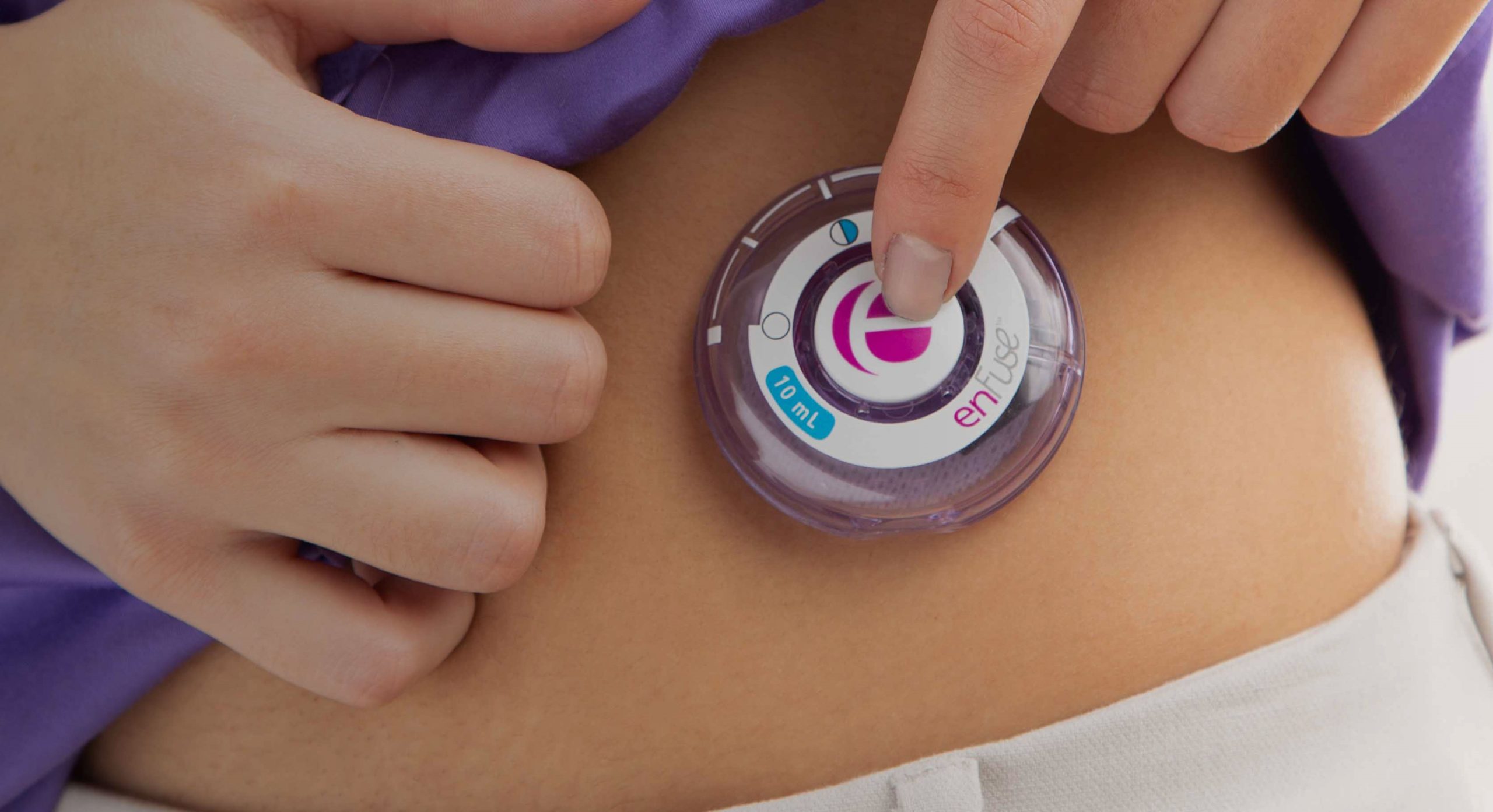
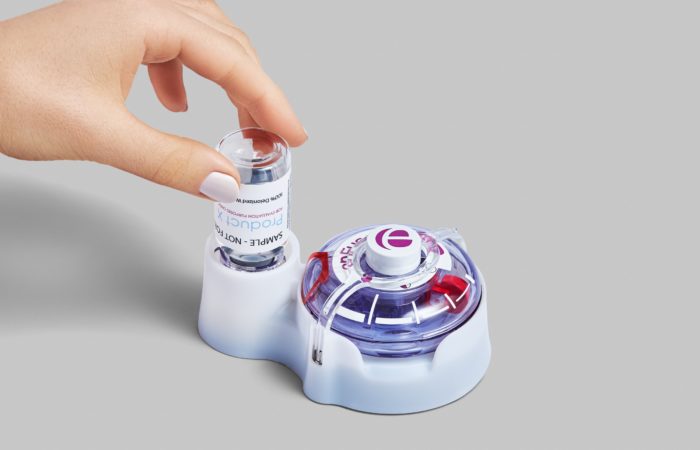
Delivery Challenges Small- and large-molecule therapeutics present unique delivery challenges for drug delivery devices. Small volumes of these therapeutics have been traditionally delivered by autoinjectors with success, up to volumes of 2.5 mL. For doses larger than 2.5 mL, traditional electromechanical infusion pumps with accompanying infusion sets have been developed but are complex and not conducive to self-administration at home. Eight of the 10 most established companies developing on-body infusors have stuck with existing electromechanical or spring-driven pump technology and simply incorporated the extension set and needle within the device. These technologies are challenged by higher volume and viscosity deliveries, as they rely on a container for the drug coupled to a power source. These systems can become problematic to wear with volumes above 10 mL. The enFuse™ device (see Box 1) uses a sequential elastomeric toroid pump, which does not require batteries to drive the pump or lights for function indicators. The container is the power source, making the enFuse efficient and small for its delivery volume.
BOX 1: THE ENFUSE ON-BODY INFUSOR
The enFuse™ platform of delivery devices is designed to enable patients to self-administer treatments at home. Moving treatment from a hospital setting to the home may potentially provide easier, more cost-effective treatments.

The enFuse is designed to deliver large volumes (up to 50 mL) of small-molecule and biologic therapeutics by the patient at home. General use on-body infusion devices able to deliver large volumes are not currently commercially available and other standard, general-use infusion pumps may require assistance from a medical professional to receive therapy at home. The enFuse technology is one of the market leaders in delivering larger volumes in a lightweight, easy-to-use device.
We believe that enabling at-home self-administration with the right wearable infusion technology can help patients can get their lives back.
Molecular Integrity
“A platform technology that can be deployed across a wide range of drug products provides incredible value to the pharma company as they don’t have to invest in developing and commercialising several different delivery solutions.”
Whatever method is used to deliver the drug, the delivery device cannot impact the integrity of the molecule being delivered. Care must be taken to ensure that whatever source it uses to deliver the drug, the function does not harm the drug substance.
Accuracy
The delivery device must also be accurate, delivering the prescribed dose as intended – no more, no less. This presents challenges for residual volume that may be left in the cartridge, container or reservoir afterdelivery. The delivery technology must account for every drop of the medication as well as ensure proper delivery into the correct anatomical space.
To accommodate for potential residual left in the device, pharma companies can overfill containers based on excess overfill guidance for viscous fluids.5 However, this overfill can be costly. The more accurate and complete the delivery, the greater the cost saving for pharma companies providing expensive therapeutics.
“It stands to reason that reducing complexity
leads to a reduction in
the potential for use errors and higher acceptance
of the technology.”
Asset Value
The ease of use and patient friendliness of the delivery technology has the potential to have a significant impact on the asset for each pharma company. If a patient population is more apt to adhere to their prescribed regimen, that regimen has a greater chance of being successful – leading to a better outcome for the patient. Additionally, for pharma companies that enter highly competitive markets or those that anticipate generic competition, an on-body infusor with high patient acceptance and adherence will differentiate its therapeutic from the competition. Lastly, a platform technology that can be deployed across a range of drug products with widely varying characteristics (e.g. delivered volume, viscosity, flow rate) provides incredible value to the pharma companies as they don’t have to invest in developing and commercialising several different delivery solutions. These true platform technologies allow pharma companies developing combination product therapeutics in conjunction with a delivery device to get to market faster with less investment and risk.
Manufacturability
The intricacy of a device, along with its manufacturability, impacts the overall price of the on-body infusor, as well as its ability to scale to market. For example, prefilled, preloaded drug delivery devices typically require aseptic assembly techniques which make manufacturing the combination product more challenging. Drug delivery device companies need to demonstrate investment in high-volume manufacturing from an early stage of development in order to benefit pharma partnerships in the long term.
Safety
Safety factors are an essential for delivery systems. As with any device, on-body infusors must operate precisely as they are engineered to function. The device and packaging need to perform as designed, and all the essential parts of the device must be sterile upon delivery to the patient.
Engineering verification, clinical studies and human factor validation studies establish the device’s safety during development. A mature quality management programme with proper document and device tracking helps to guide the development and launch of delivery devices and respond to potential issues post-market.
PATIENT CHALLENGES
Living with a medical condition is difficult and travelling to a healthcare facility for IV infusions makes life even more challenging. But with the promise of self-administration treatments at home, a patient can get part of their life back.
Size
One prohibitive factor for patients is the size of the device itself. For patients to use an on-body infusor, the device must be comfortable and easy to handle, especially for those who have dexterity and mobility challenges.
The size of a device grows quickly when pumps, batteries and traditional vials or cartridges have to be incorporated. The larger the device, the heavier it becomes, and the more difficult it can be to ensure it remains adhered to a patient’s skin at the intended location for the duration of the infusion. A lighter, less bulky infusor allows the patient to have a more comfortable experience. In addition, a more discreet delivery device permits the user to go about their normal activities as they wear the infusor.
Steps
It stands to reason that reducing complexity leads to a reduction in the potential for use errors and higher acceptance of the technology. Device designers must take into account the number of steps a patient must execute in order to administer their treatment. Human factors testing helps to elucidate potential failure modes and ensure the overall design and instructions for use are adequate. However, the number of steps required may not be sufficient in assessing the overall risk of error; the complexity of those steps must also be assessed.
Most on-body infusors in development have defaulted to using prefilled cartridges which are often already preloaded into the infusion device for the patient. Using these devices requires multiple steps by the user that may not be obvious. Often this begins by removing the device from cold storage and allowing it to warm to room temperature which could take up to an hour.6 Notwithstanding the injection tolerability of a cold drug, some of these devices will not perform properly due to the high viscosity of the cold drug. While a patient-filled and loaded device requires an additional step to load the device, this may be preferred to waiting to start the infusion, especially if it could be used immediately after loading. The challenge for designers is to ensure loading the device is as simple as possible.
To aid the patient in self-administration, companies like Enable Injections are developing training devices. In addition, Bluetooth connectivity options can assist the patient in steps required for administration, as well as prompt convenient reminders for date and time for administration.
Digital
“A platform technology that can be deployed across a wide range of drug products provides incredible value to the pharma company as they don’t have to invest in developing and commercialising several different delivery solutions.”
With Bluetooth connectivity built into an on-body infusor, caregivers and otherpeople involved in a patient’s care can remotely monitor a patient’s infusion. Real-time data can be transmitted in a low-energy signal and can often offer proof that the infusion was performed, as well as provide other meaningful data. With appropriate permissions given, digital data transmitted by Bluetooth connectivity can provide information to healthcare providers and payers – and could provide a means of confirming adherence.
Cost
Decreased healthcare deductibles could provide extra incentive for patients to perform their infusions at home via on-body infusors. By administering at home, patients can decrease their healthcare expenses and administration costs, as well as eliminate travel to and from a healthcare facility for infusions and the disruptions to normal life routines including school and work missed due to IV infusion.
Safety
Many patients experience a psychological burden of undergoing treatment involving any type of needle, including IV needles, autoinjector needles and prefilled syringes. An exposed needle any time during the administration can be an intimidating factor for many patients. With a well-designed on-body infusor, the patient does not need to be exposed to the needle at any time before, during or after the infusion, which may alleviate the psychological burden and enable them to adhere to treatment.
Patient Value
By reducing travel, the risk of acquiring an infection in a clinic and the cost of healthcare administration of IV therapeutics for many disease states, on-body infusors have the potential to provide value and improve quality of life for the patient.
THE IMPACT OF WEARABLE TECHNOLOGY
Currently, delivery of large-volume biologic and small-molecule therapeutics is inconvenient, expensive and difficult. However, new technology developments with on-body infusors which can deliver large volumes safely and conveniently in the home can change the treatment paradigm and create an improved patient experience. Patients will benefit, and pharma companies will be able to differentiate products through a patient-preferred, on-body infusor platform.
Enable Injections is one such company developing and manufacturing large-volume on-body infusors, with the goal of moving patients out of the clinic, reducing the cost of healthcare and improving patient quality of life.
REFERENCES
- Kamal R et al, “How much is health spending expected to grow?”. Peterson-Kaiser Health System Tracker, 2019.
- Schmidt B, “Home Infusion Therapy: Safety, Efficacy, and Cost-Savings”. PSQH, 2012.
- Beijer S, “Treatment adherence and patients’ acceptance of home infusions with adenosine 5’-triphosphate (ATP) in palliative home care”. Support Cancer Care, 2008, Vol 16(12), pp 1419-1424.
- “Subcutaneous Biologics, Technologies and Drug Delivery Systems 2018-2030 (2nd Edition)”. Research Report, Roots Analysis, 2018, pp 243.
- “Allowable Excess Volume and Labeled Vial Fill Size in Injectable Drug and Biological Products”. US FDA, 2015.
- Prescribing Information for Repatha (evolocumab) injection, Amgen. February 2019.